 Purpose: Double-wave diffusion experiments offer the possibility of probing correlation between molecular diffusion at multiple time points. It has recently been shown that this technique is capable of measuring the exchange of water across cellular membranes. The aim of this study was to investigate the effect of macroscopic tissue anisotropy on the measurement of the apparent exchange rate (AXR) in multicompartment systems.
Purpose: Double-wave diffusion experiments offer the possibility of probing correlation between molecular diffusion at multiple time points. It has recently been shown that this technique is capable of measuring the exchange of water across cellular membranes. The aim of this study was to investigate the effect of macroscopic tissue anisotropy on the measurement of the apparent exchange rate (AXR) in multicompartment systems.
Method: AXR data were collected from a perfusion-fixated vervet monkey brain tissue at high angular resolution on a preclinical imaging system. To analyze the effect of tissue anisotropy the AXR was expanded for anisotropic systems by calculating scalar AXR values along the principal directions of the diffusion tensor.
Results: In fixated brain tissue, the measured AXR was sensitive to the orientation of anisotropic structures. AXR, especially in white matter, was robustly estimated along the first and second principal directions of the diffusion tensor, but increasing noise was seen in the AXR estimates along the third principal direction of the diffusion tensor.
The dataset made available for download was used in Sønderby et al 2013 where further details of the study can be found.
MATLAB code for reproducing the results are described below and furthermore supplementary simulations not found in the paper supporting our conclusions are presented.
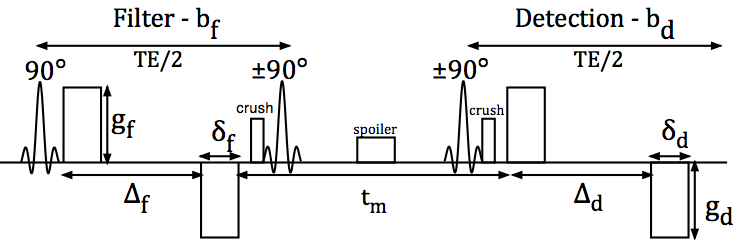 |
| FIG. 1 (Sønderby et. al. 2013): Timing diagram of the double bipolar stimulated echo sequence used in this study. The two sets of diffusion-encoding gradients, denoted the filter block and detection block, are separated by a variable mixing time tm. During echo time (TE), the signal is subject to T2 relaxation, whereas only T1 relaxation is experienced between the second and third 90° RF pulses due to z-storage of the magnetization. The spoiler gradient dephases any transversal magnetization excited by the second 90° RF pulse. The crusher gradients remove unwanted signal pathways due to imperfect RF pulses but also introduce diffusion weighting in the slice-encoding direction across tm. © Wiley Periodicals, Inc. |
 |
|
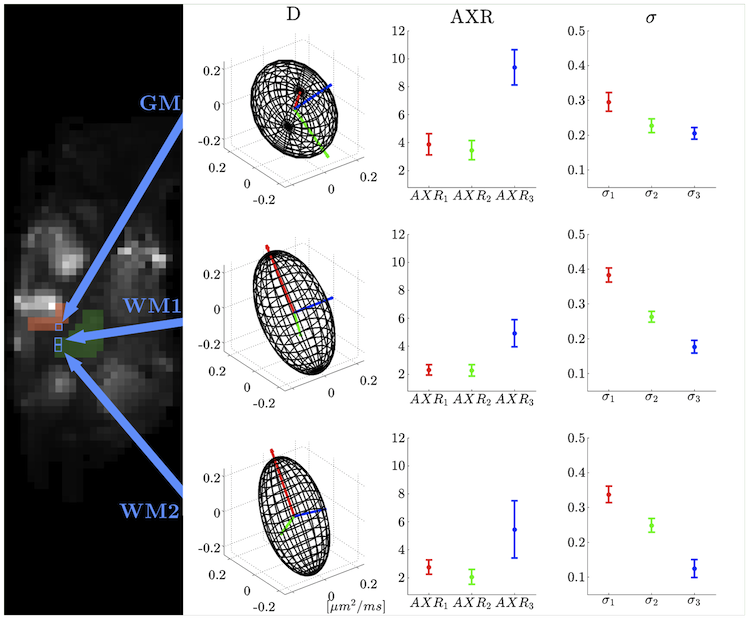
FIG. 3 (Sønderby et. al. 2013): Illustration of (left) the sagittal b0 slice plane from the left hemisphere, slightly more lateral than the caudate nucleus, and (right) diffusion tensor D, AXR, and filter efficiency &sigma. Red, green, and blue indicate the first, second, and third principal directions of D, respectively. The top row illustrates tensors from a voxel in GM. The middle and bottom rows illustrate tensors from two voxels in WM. Arrows indicate voxel positions. © Wiley Periodicals, Inc. |
Dataset description:
All experiments were performed in an experimental 4.7 T MRI scanner (Agilent Technologies Inc., Santa Clara, California) with a maximal gradient strength of 400 mT/ m. A parasagittal section from a vervet monkey brain from the Montreal brain bank, prepared as described by Dyrby et al. 2011 was positioned on top of a homemade 20-mm surface coil and placed in an RF transmit coil (RAPID Biomedical GmbH, Rimpar, Germany). The monkey was handled and cared for on the island of St. Kitts (The Caribbean Primate Center of St. Kitts, West Indies) according to a protocol approved by the local ethics committee.
The data was acquired with a Filter Exchange Imaging sequence (FEXI) with the following parameters were used for all acquisitions: TR: 2500 ms, TE: 43 ms, bfilter = 3148 s/mm2 (gradient strength [G] = 320 mT/m, pulse duration [delta] 7 ms, pulse separation [DELTA] 11.1 ms), and bdetection = 3050 s/mm2 (G = 320 mT/m, delta = 8.3 ms, DELTA = 48.8 ms). The mixing time (tm) was varied in seven steps [9, 57, 106, 145, 203, 251, 300] ms For ADC estimation, a single wave measurement with bfilter = 0 s/mm2 and bdetection = 3050 s/mm2 was acquired. A voxel size of 1 x 1 x 1.5 mm3 and four repetitions ensured a lowest SNR >30 in WM with all gradients aplied. The temperature was 20.2±0.4°C during the entire experiment.
For each tm the FEXI protocol was repeated with 20 uniformly distributed gradient directions from the Camino software.
Using these data the normal diffusion tensor D and and the mixing time dependent tensors D'(tm) (for each tm) was calculated in each voxel, i.e.:
- Estimating D from a diffusion-weighted signal where only the detection block gradients were applied.
- Estimating D’(tm) from diffusion-weighed signals, where both the filter and detection block gradients were applied separated by varying mixing times as described above.
- AXRn and sigman were obtained by fitting Eq. [4] in Sønderby et al 2013 to D and D’(tm) (See MATLAB scripts below).
Data Download and Analysis
Step-by-step Guide
- Download the dataset in NifTI format from here. The Zip-file contains the matlab files for analyzing the data
- Extract the Zip-file to (folder: AXR_dataset)
- Download the Tools for NIfTI and ANALYZE image MATLAB package. Extract the content, rename the folder to NIFTI and move it to the AXR_dataset folder.
- Run the AXR_analysis.m scripts in the AXR_dataset folder
Simulations
To address the effect of micro-structural anisotropy we have included Monte Carlo simulations of an anisotropic environments consisting of empty cylinders and spheres with different orientation and permeability as seen in Figure A and Figure B.
 |
 |
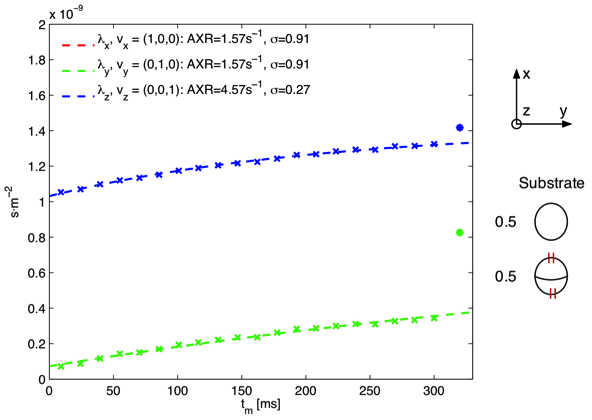
| Figure A: Monte Carlo simulation in a substrate consisting of impermeable axons parallel to the z-axis and permeable spheres. The eigenvalues D of D’ are shown as asterisks and crosses respectively. Note that the λ_x(red) and λ_y(green) data points are identical leaving only the λ_y data points visible. |
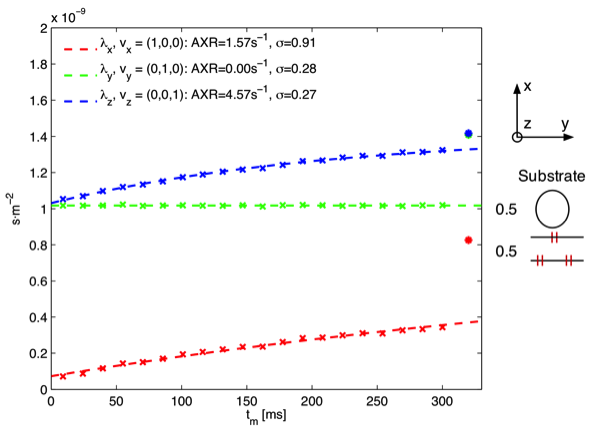
Figure B: Monte Carlo simulation in a substrate consisting of impermeable axons parallel to the z-axis and permeable axons parallel to the y-axis. The eigenvalues of D and D’ are shown as asterisks and crosses respectively.
|
In the simulation of impermeable axons parallel to the z-axis and permeable spheres, seen in Figure A, the highest AXR is measured in the z-direction parallel to the impermeable axon i.e. AXR is modulated by an anisotropic compartment not engaged in exchange processes. Previous studies have assumed that the anisotropy should only be reflected in the filter efficiency (Nilsson et al 2012). Furthermore we note that low filter efficiency corresponds to high AXR, which was also observed in our experimental data. In Figure B a simulation of impermeable axons parallel to the z-axis and permeable axons parallel to the y-axis is seen. Similar to Figure A the measured AXR is modulated by the impermeable anisotropic compartments and furthermore we note that no exchange is observed when measuring parallel to the (empty) permeable axons as expected
Based on the above observations we believe that tissue anisotropy is at least partly responsible for the observed ‘anisotropy’ of the measured AXR. However a simple cylinder model as used in the simulation, cannot explain the reliable estimation of AXR in directions with fast diffusion as indicated by the data presented in the article, since cylinders parallel to the gradient directions are not “visible” and hence not available for exchange in these models. These findings indicate that the measured AXR depends on membrane permeability but also other factors such as tissue anisotropy and possibly intracellular structures (not taken into account in our simple simulation model) in a complex fashion which is subject to further studies
Please cite the following papers
1) Describing the new theory, method and how the data was acquired:
Sønderby, C. K., Lundell, H. M., Søgaard, L. V. and Dyrby, T. B. (2013), Apparent exchange rate imaging in anisotropic systems. Magn Reson Med. doi: 10.1002/mrm.24957
2) Describing the preparation of the vervet monkey brain tissue:
Dyrby TB, Baaré WFC, Alexander DC, Jelsing J, Garde E, Søgaard LV. An ex vivo imaging pipeline for producing high-quality and high resolution diffusion-weighted imaging datasets. Hum Brain Mapp 2011;32:544–563
Observed problems or artifacts: None
Funding
The EU founded CONNECT consortium under the FET program, The Lundbeck Foundation and the Otto Mønsted Foundation
References
Nilsson, M., Lätt, J., van Westen, D., Brockstedt, S., Lasič, S., Ståhlberg, F., & Topgaard, D. (2012). Noninvasive mapping of water diffusional exchange in the human brain using filter‐exchange imaging. Magnetic Resonance in Medicine.


