PRECISION NEUROIMAGING OF BRAIN DISORDERS
The field of Magnetic Resonance Imaging (MRI) continues to advance, expanding the possibilities to study how brain disorders affect the brain´s structure, function, and metabolism. We exploit the enormous biomedical potential of MRI to establish MR-based Precision Medicine as a powerful interface between diagnostic radiology and the clinical neurosciences.
Our goal is to identify clinically relevant MR-based markers that provide relevant information about the individual risk or prognosis with respect to the course of disease or to response to treatment. This will boost the role of MRI as a clinical tool that can help neurologists, neurosurgeons, and psychiatrists to tailor their therapy to the individual needs of their patients.
Embedded in a large University Hospital, researchers at DRCMR have long-standing collaborations with numerous clinical research groups in the Capital Region of Denmark. Our clinical neuroimaging research covers a wide range of psychiatric and neurologic disorders as well as the entire life span. The Clinical Precision Imaging area is closely intertwined with and capitalizes on the specific expertise and outcomes of the MR Physics & Analyses, Precision Brain Stimulation, Cognitive & Computational Neuroscience and Life Span Imaging research areas.
RESEARCH STRATEGY
We develop and validate novel MR-based technologies which can reliably capture risk, resilience or disease-related structural, functional, metabolic changes in the brain. In parallel, we develop and advance neuroimaging data-analysis tools. MR-based imaging technologies and analytical tools are optimized to reliably identify clinically relevant MR-based “read-outs”. It is our aim to further optimize the value of MR-based precision medicine by integrating longitudinal electronic health data (E-health) and genomic and biochemical data (“Omics”). Another line of our research is concerned with exploiting MRI-based techniques to individualize the use of non-invasive transcranial brain stimulation (NTBS) in order to maximize therapeutic efficiency.
BRIDGING MR-BASED NEUROIMAGING, NEUROSCIENCE AND CLINICAL PRACTICE
Our research will yield novel MR protocols that pick-up disease-causing (pathogenesis) or disability-causing (pathophysiology) processes at high sensitivity and specificity. It is our aim to establish predictive MR-based neuroimaging markers (“read outs”) to assist clinical decision making and personalized medicine. Another aim is to identify pre-symptomatic MRI-based neuroimaging markers of disease formation in cohorts bearing increased risk or resilience (reduced risk) for a neurologic (e.g. Parkinson´s disease) or psychiatric (e.g. schizophrenia) disorder and in large population cohorts. Finally, we wish to establish novel MRI-based neuroimaging markers for the presence or magnitude of common disabling symptoms that can be used to test and monitor the efficiency of symptomatic treatments at the individual level.
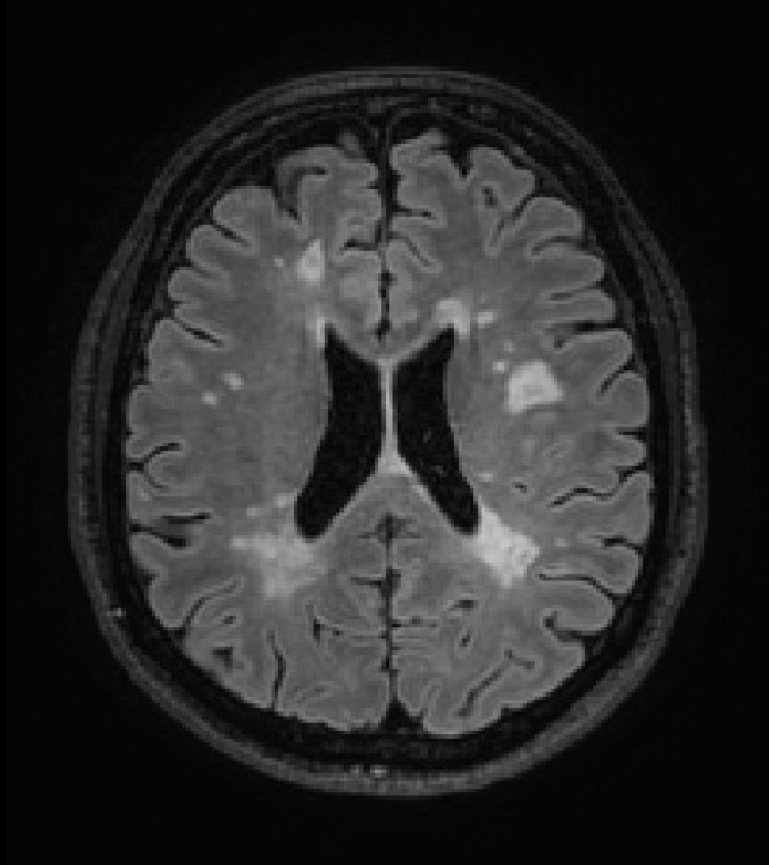
Axial Magnetic Resonance Image (MRI) showing characteristic disease-related white matter alterations in a patient with multiple sclerosis



