We as humans are proficient in predicting the perceptual, social or emotional outcome of our actions because our brain has strong predictive associations about action/outcome contingenices at its disposal. In recent years, substantial progress has been made in identifying neural mechanisms involved in updating internal representations of action-outcome contingencies. Below are three examples of studies on prediction and learning performed in the ContAct group.
Study 1
Hulme OJ, Skov M, Chadwick MJ, Siebner HR, Ramsøy TZ (2014) Sparse encoding of automatic visual association in hippocampal networks. NeuroImage 102, Part 2:458–464.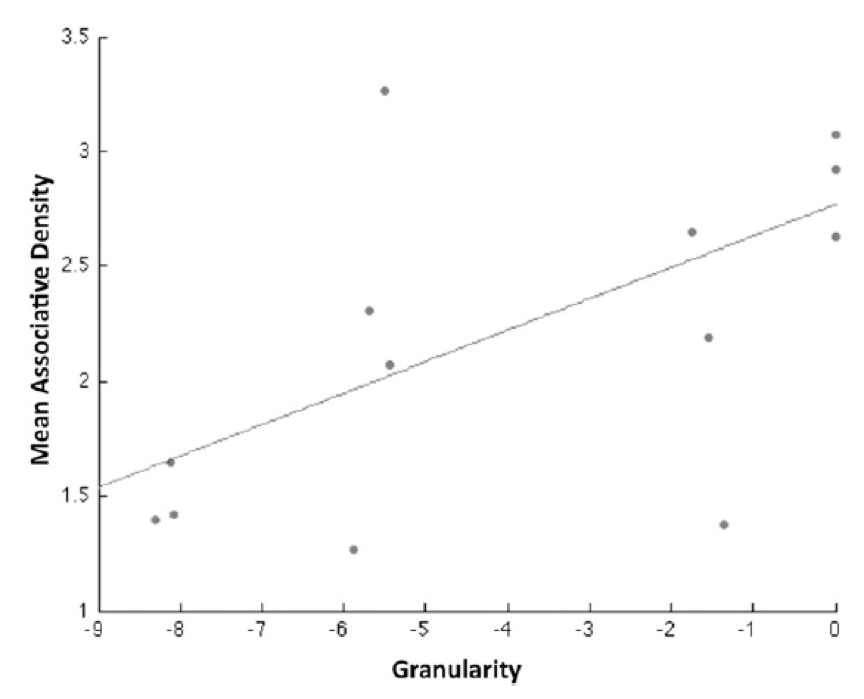
This paper pertains to WP4 Prediction and Learning, as it uses fMRI and Bayesian Decoding Methods to reveal the nature of associative predictions and the sparsity of their neuronal representation in the medial temporal lobe. The hippocampus and mediotemporal cortex are endowed with the network topology, physiology, and neurochemistry to automatically and sparsely code sensori-cognitive associations that can be reconstructed from single or partial inputs. Whilst acquiring fMRI data and performing an attentional task, participants were incidentally presented with a sequence of cartoon images. By assigning subjects a post-scan free-association task on the same images we assayed the density of associations triggered by these stimuli. Using multivariate Bayesian decoding, we show that human hippocampal and temporal neocortical structures host sparse associative representations that are automatically triggered by visual input. Remarkably, the sparsity of CA encoding correlated significantly with associative memory performance over subjects.
The figure on the right shows intersubject plot of evidence for the for the sparse model compared to a clustered model in the left CA subfield (mean log odds) vs. associative memory performance.
Study 2
Meder D, Madsen KH, Hulme O, Siebner HR (2016) Chasing probabilities — Signaling negative and positive prediction errors across domains. NeuroImage 134:180–191.
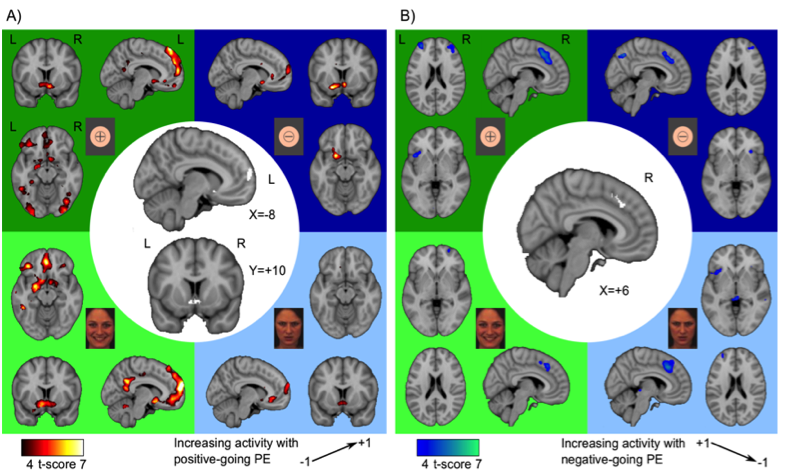
Prediction errors (PEs), the surprise about an outcome of an action, inform choice behavior across a diversity of outcome domains and dimensions, yet it is unclear whether their neural correlates pertain to computational features that are invariant with respect to outcome valence, perceptual sensory domain, or some combination of the two. We used four learning tasks which differed in terms of contextual outcome valence (reward-seeking versus punishment-avoidance) and domain (abstract symbols versus facial expressions) of outcomes. We found that three areas that coded PEs in all conditions (see figure). Outcome valence influenced PE-related activity in left amygdala, IFG, and dorsomedial prefrontal cortex, where activity selectively scaled with increasingly positive PEs in the reward-seeking but not punishment-avoidance context, irrespective of sensory domain. Left amygdala displayed an additional influence of sensory domain. In the context of avoiding punishment, amygdala activity increased with increasingly negative PEs, but only for facial stimuli, indicating an integration of outcome valence and sensory domain during probabilistic choices.
This figure shows brain regions where outcome related activity scaled to positive-going (A) and negative-going PEs (B). The scaling of regional activity is also given for each condition separately (green and blue squares). Top left (dark green): Reward-Abstract condition. Top right (dark blue): Punishment-Abstract. Bottom left (light green): Reward-Faces. Bottom right (light blue): Punishment-Faces. Ventral striatum and frontopolar cortex coded increasingly positive PEs in all four conditions, whereas dorsal anterior cingulate cortex (dACC) traced increasingly negative PEs in all conditions.
Study 3
Vegt JPM van der, Hulme OJ, Zittel S, Madsen KH, Weiss MM, Buhmann C, Bloem BR, Münchau A, Siebner HR (2013) Attenuated neural response to gamble outcomes in drug-naive patients with Parkinson’s disease. Brain 136:1192–1203.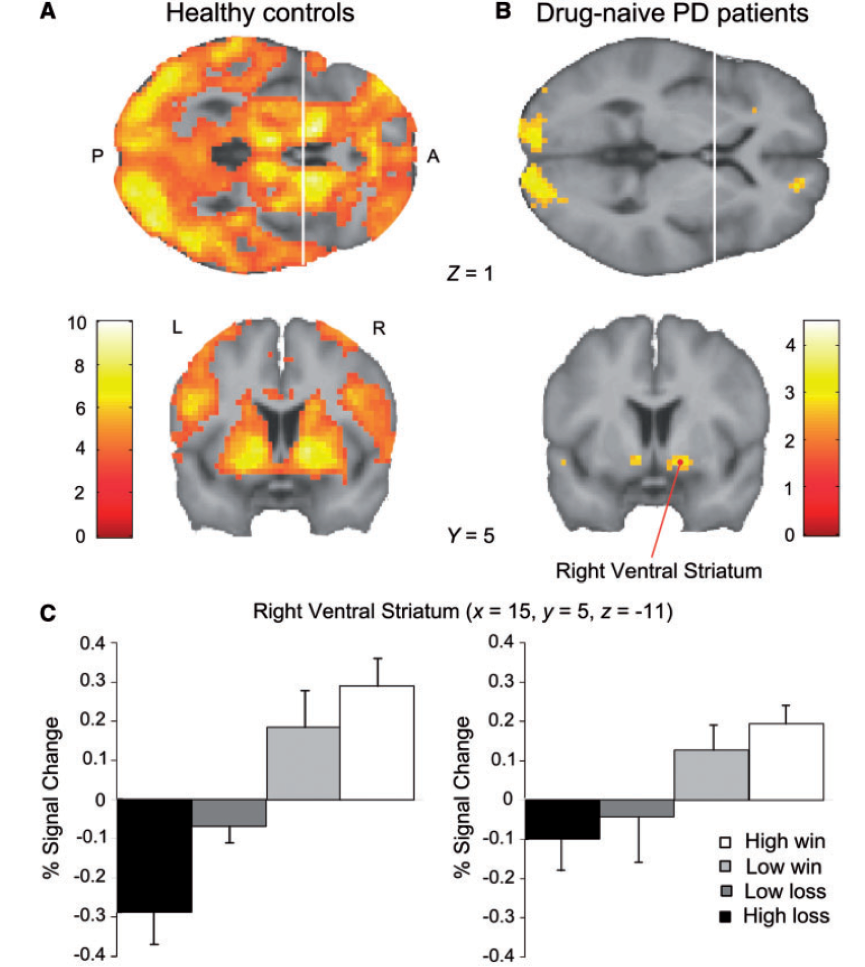
Parkinson’s disease not only leads to motor impairment bu also affects reward processing, but disease-related deficits in reinforcement learning are thought to emerge at a slower pace. We used fMRI to investigate brain activity in Parkinson’s patients and control subjects as they performed a simple two-choice gambling task resulting in monetary gains and losses. In patients with Parkinson’s disease, the neural response to reward outcome was attenuated in a large group of mesolimbic and mesocortical regions. The results show that the core regions of the meso-cortico-limbic dopaminergic system, including the ventral tegmental area, ventral striatum, and medial orbitofrontal cortex, are already significantly compromised in the early stages of the disease and that these deficits cannot be attributed to the contaminating effect of dopaminergic treatment.
The figure on the right shows the effect of outcome value on neural activity in healthy subjects (A) and Parkinson’s patients (B). (C) Activation profile of outcome-related activity in right ventral striatum. Healthy controls (left) show a stronger increase in outcome-related activity with outcome value than patients with Parkinson’s disease.


